
Spetic holds a small tomato. Photograph by Russell Lee, courtesy of CWRU.
Researchers with the Advanced Platform Technology (APT) Center at Louis Stokes Cleveland VA Medical Center (Cleveland VA), Ohio, and at Case Western Reserve University (CWRU), Cleveland, have found a way to provide sensory feedback for a prosthetic hand via a direct interface to the residual peripheral nerve. The results of their most recent study show that the implanted peripheral nerve interfaces in two human subjects with upper-limb amputation provided stable, natural touch sensation in their hands for more than one year. The results were published October 8 in the Science Translational Medicine journal. To watch a video of the research, click here.
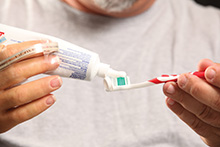
Vonderhuevel squeezes toothpaste onto a toothbrush. Photograph by Dale Omori, courtesy of CWRU.
Two patients underwent the procedure. Surgeons Michael W Keith, MD, and J. Robert Anderson, MD, from the CWRU School of Medicine and the Cleveland VA, implanted three electrode cuffs in Igor Spetic’s forearm 30 months ago, enabling him to feel 19 distinct points; and two cuffs were implanted in Keith Vonderhuevel’s upper arm 18 months ago, enabling him to feel 16 distinct locations. Despite having phantom limb pain, both men said that the first time they were connected to the system and received the electrical stimulation was the first time they felt their hands since their respective accidents. In the ensuing months, they began feeling familiar sensations and were able to control their prosthetic hands with more dexterity. Further, Spetic and Vonderhuevel each said their phantom limb pain has subsided substantially.
The system, which is limited to the lab at this point, uses electrical stimulation to give the sense of feeling. But there are key differences from other reported efforts. First, the nerves that used to relay the sense of touch to the brain are stimulated by contact points on cuffs that encircle major nerve bundles in the arm, not by electrodes inserted through the protective nerve membranes. Second, when they began the study, the sensation Spetic felt when a sensor was touched was a tingle. To provide more natural sensations, the research team has developed algorithms that convert the input from sensors taped to a patient’s hand into varying patterns and intensities of electrical signals. The sensors themselves aren’t sophisticated enough to discern textures, they detect only pressure. Third, the system has worked in excess of a year and a half. Other research has reported sensation lasting one month and, in some cases, the ability to feel began to fade over weeks.
“The sense of touch is one of the ways we interact with objects around us,” said Dustin Tyler, PhD, an associate professor of biomedical engineering at CWRU and director of the research. “Our goal is not just to restore function, but to build a reconnection to the world. This is long-lasting, chronic restoration of sensation over multiple points across the hand.
“The work reactivates areas of the brain that produce the sense of touch, said Tyler, who is also associate director of the Advanced Platform Technology Center at the Cleveland VA. “When the hand is lost, the inputs that switched on these areas were lost.”
“The sense of touch actually gets better,” said Vonderhuevel, who lost his hand in 2005 and had the system implanted in January 2013. “They change things on the computer to change the sensation. One time it felt like water running across the back of my hand.”
The different signal patterns, passed through the cuffs, are read as different stimuli by the brain. The scientists continue to fine-tune the patterns, and Spetic and Vonderhuevel appear to be becoming more attuned to them. A blindfolded Vonderhuevel has held grapes or cherries in his prosthetic hand-the signals enable him to gauge how tightly he’s squeezing-and pulled out the stems. “When the sensation’s on, it’s not too hard,” he said. “When it’s off, you make a lot of grape juice.”
Different signal patterns interpreted as sandpaper, a smooth surface, and a ridged surface enabled a blindfolded Spetic to discern each as they were applied to his hand. And when researchers touched two different locations with two different textures at the same time, he could discern the type and location of each.
Tyler believes that everyone creates a map of sensations from their life history that enables them to correlate an input to a given sensation. “I don’t presume the stimuli we’re giving is hitting the spots on the map exactly, but they’re familiar enough that the brain identifies what it is,” he said.
Because of Vonderhuevel’s and Spetic’s continuing progress, Tyler is hopeful the method can lead to a lifetime of use. He’s optimistic that within five years his team can develop a system for a patient to use at home. In addition to prosthetic hands, Tyler believes the technology can be used to help those using prosthetic legs receive input from the ground and adjust to gravel or uneven surfaces.
Editor’s note: This story was adapted from materials provided by Case Western Reserve University.




