University of California, Los Angeles (UCLA) researchers, led by Heather Maynard, PhD, an associate professor of chemistry and biochemistry, and a member of UCLA’s California NanoSystems Institute, are developing a new biomimicry healing technique to treat skin wounds, such as diabetes-related foot ulcers. The results of their research were published in the February online edition of the journal Nature Chemistry.
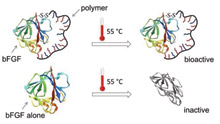
When subjected to normal conditions bFGF may encounter during transport, storage, or therapeutic use, the bFGF-heparin mimicking conjugate remains active, while natural bFGF no longer has any biological activity.
Photograph by Thi Nguyen, courtesy of UCLA.
Among the key players involved in natural wound healing is a signaling molecule called basic fibroblast growth factor (bFGF), which is secreted by cells to trigger healing processes, as well as embryonic development, tissue regeneration, bone regeneration, the development and maintenance of the nervous system, and stem cell renewal. Since the importance of bFGF was recognized in 1973, researchers have applied bFGF to wounds, but the treatments have not been very effective. What scientists now recognize, Maynard said, is that these growth factors typically lose their activity quickly in storage.
Using the principle of mimicry, Maynard and her team discovered how to stabilize bFGF, making it more suitable for medical applications. Relying on bFGF’s ability to bind heparin-a naturally occurring complex sugar found on the surface of our cells-the team synthesized a polymer that mimics the structure of heparin. Their new polymer, p(SS-co-PEGMA), mimics heparin’s natural ability to stabilize the growth factor. When attached to bFGF, the new polymer makes the protein stable to the stresses that normally inactivate it, making it more suitable for medical applications. After showing that p(SS-co-PEGMA) was non-toxic to the human cells important in wound healing, the research team used it to conjugate bFGF and demonstrated that the growth factor could remain active outside of the body for extended periods of time, even after it is exposed to heat, cold, damaging enzymes that would normally break it down, and acidic conditions like those found in wound injuries.
Moreover, they showed that this bound bFGF functions just like normal bFGF to trigger the same signaling pathways involved in the healing process.
The ability to stabilize bFGF means that it can be potentially stored, shipped, and made available for use by doctors and patients as needed, Maynard said.
The new material is now being tested by dermatologists. The team is also researching ways to stabilize other proteins involved in wound healing and ways to make bFGF more active.
Editor’s note: This story was adapted from materials provided by UCLA.





