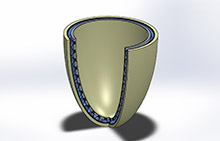
Image of the bubble actuator, embedded in a liner, that the UT Arlington research team is designing and building, courtesy of UT Arlington.
University of Texas at Arlington (UT Arlington) researchers have been awarded a $744,300 grant from the U.S. Department of Defense Peer Reviewed Orthapaedic Research Program to create an adaptive interface that fits between a prosthesis and a patient’s limb so that the fit and comfort of the prosthesis are improved.
Haiying Huang, PhD, an associate professor in the Mechanical & Aerospace Engineering Department, and Muthu Wijesundara, PhD, a principal research scientist at UT Arlington’s Research Institute, are collaborating on the project. The interface will resemble an inflatable bubble wrap that will be embedded with sensors. Huang is an expert in sensor technology. She said four types of sensors will be used with the interface to monitor the fit of the prosthetic device. The sensors will measure vertical movement of the limb relative to the socket wall, the pressure on the limb, changes in the circumference of the residual limb during the day, and the water content in the tissue.
“Eventually, we want to build the socket that can adjust automatically to the patient,” Huang said. “In order to do that, we need the sensors to tell us when and how to adjust the socket. We plan to design a warning system first, then the sensor data will teach us how to adjust the interface automatically.”
Currently, many prosthetic users manually adjust the fitting between their prostheses and their limbs by adding or removing socks.
Wijesundara, who specializes in medical devices for applications in tissue regeneration, wound healing, and prosthetic devices/interfaces, said they want an adaptable interface that can improve comfort and the fit regardless of the residual limb conditions and improve the user’s the quality of the life. “We want everything to adjust depending on whether the person is walking, running, or simply sitting down,” Wijesundara said. “This interface technology can be applied to various prosthetic devices and exoskeleton applications.”
The researchers said they believe it could take three to five years to start clinical applications of the device.
Editor’s note: This story was adapted from materials provided by UT Arlington.





