A team of researchers conducted what they said is the first meta-analysis of the available published research on the clinical effectiveness and safety of powered exoskeletons in patients with spinal cord injuries (SCI). They searched the MEDLINE and EMBASE databases for studies of powered exoskeleton-assisted walking in patients with SCIs. They found that, following the exoskeleton training program, 76 percent of patients were able to ambulate with no other physical assistance.
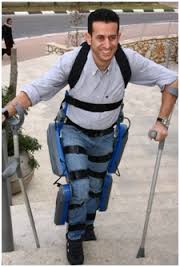
Photograph of the ReWalk courtesy of Argo Medical Technologies.
A total of 14 studies (eight of the ReWalk Robotics ReWalk™, three of the Ekso Bionics Ekso™, two of the Parker Hannifin Indego®, and one unspecified exoskeleton) representing 111 patients were included in the analysis. Training programs were typically conducted three times per week, 60-120 minutes per session, for one to 24 weeks. Ten studies used flat indoor surfaces for training and four studies incorporated training that including walking outdoors, navigating obstacles, climbing and descending stairs, and performing activities of daily living. The weighted mean distance for the six-minute walk test was 98m. The physiologic demand of powered exoskeleton-assisted walking was 3.3 metabolic equivalents and rating of perceived exertion was 10 on the Borg 6-20 scale, comparable to self-reported exertion of an able-bodied person walking at three miles per hour. Improvements in spasticity and bowel movement regularity were reported in 38 percent and 61 percent of patients, respectively. No serious adverse events occurred. The fall incidence at any time during training was 4.4 percent, all occurring while tethered using a first-generation exoskeleton and none resulting in injury. The incidence of bone fracture during training was 3.4 percent. These risks have since been mitigated with newer generation exoskeletons and refinements to patient eligibility criteria, the researchers said. They concluded that the test results indicate that powered exoskeletons allow patients with SCIs to safely ambulate in real-world settings at a physical activity intensity conducive to prolonged use and known to yield health benefits.
The study, “Clinical Effectiveness and Safety of Powered Exoskeleton-assisted Walking in Patients With Spinal Cord Injury: Systematic Review With Meta-analysis,” was published March 22 in the open access journal Medical Devices: Evidence and Research. ReWalk Inc., Yokneam Ilit, Israel, and Marlborough, Massachusetts, disclosed that it provided support for the study, but had no editorial control, and the authors reported no conflicts of interest.




