It all started with a rabbit.
In 1952, Prof. Per-Ingvar Branemark of Sweden used a titanium implant chamber to study blood flow in rabbit bone. At the end of the experiment he discovered that the bone had integrated so completely with the implant that the chamber could not be removed. Branemark called the discovery “osseointegration,” and saw the possibilities for human use. The procedure was first implemented in dentistry for fixation of teeth and now also is used for cranial and maxillofacial reconstruction as well.
Since 1990 the procedure has been used in lower- and
upper-extremity prosthetic treatment. The pioneer limb
osseointegration patient had very high bilateral transfemoral
amputations, making the use of traditional prostheses very
difficult, if not impossible. She was implanted with
osseointegrated prostheses, and is now married with two daughters
and doing well, Rickard Branemark noted in a presentation at Walter
Reed Army Medical Center and Department of Veterans Affairs (VA)
Amputee Healthcare & Prosthetics Workshop November 17-18,
2003.
Benefits, Challenges
Amputees report several advantages over
conventional prostheses:
- Less feeling of weight;
- More control over the prosthesis;
- No perspiration, pain, and tissue breakdown from an external
socket; - No need to remake a socket;
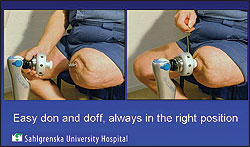
- Easier donning and doffing; and
- “Osseoperception”–the term given to the patient-reported
feeling of heightened perception of the environment with
osseointegrated prostheses.
Research suggests osseoperception is secondary to nerve ingrowth into remodeling bone, as controlled by neuropeptides such as calcitonin gene-related peptide, Rickard Branemark noted. Osseoperception has been quantified using vibrametry as a measure of neural sensory function, as reported in Prosthetics Orthotics International, 2000. There’s intriguing ongoing research work on osseoperception in Sweden and at the VA Healthcare Center, San Diego, and the University of California-San Diego focusing on the relationship between nerve injury and remapping of the
somatosensory cortex.
The current disadvantages of osseointegration are:
- Two surgeries required;
- Relatively long rehabilitation period; and
- Deep-infection risk. The risk of infection can be largely
controlled through rigorous personal hygiene and antibiotics. If
infection does occur, it can cause major problems: bone loss,
loosening of the implant, and a possible need to re-amputate the
limb at a higher, less functional level.
However, as a team from the Department of Prosthetics and
Orthotics, Sahlgren University Hospital, Goteborg, Sweden,
explained during a presentation at the 2006 Annual Meeting of the
American Academy of Orthotists and Prosthetists (the Academy),
today a strict protocol called OPRA (Osseointegration of Prostheses
for Rehabilitation Amputees) is followed that has greatly reduced
deep infection and implant loosening. Although the first years
after 1990 saw a 40-percent loosening/deep infection rate, new
treatment protocols have yielded an 85-percent success rate for
osseointegrated prosthetic treatment, according to the team. Since
1999, the success rate has been higher than 95 percent of patients
who have passed their two-year follow-up mark.
Treatment Procedure
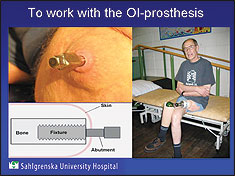
Now used more widely in Europe, osseointegration involves
threading the prosthesis onto a titanium bolt, called an
“abutment,” which is implanted in the bone of the residual limb and
protrudes through the skin. The abutment provides the attachment
for the prosthesis. The treatment involves two surgical procedures,
explained the Sahlgren University Hospital team–orthopedic
engineers Stewe Jonsson and Eva Haggstrom and occupational
therapist Kerstin Caine-Winterberger. During stage one the fixture
is threaded into the skeleton, the wound is completely closed, and
the healing period starts, lasting normally about six months. The
implant is unloaded until the bone has grown into the threads; a
temporary socket prosthesis can be used during this period.
At the second stage, the implanted fixture is re-exposed and the
abutment is connected to the fixture; the wound is closed with the
abutment penetrating the skin. The final phase is mobilization,
during which the implant is gradually loaded by using a training
prosthesis. Depending on amputation level, bone quality, and other
factors, the mobilization time can take up to six months and
possibly longer, according to the team.

About 100 patients currently have undergone lower-extremity osseointegration treatment, the majority of them transfemoral amputees.
About 30 patients have undergone osseointegration procedures for both transhumeral and transradial amputees, with implants being used in both radius and ulna for the transradial patients. Different types of prostheses can be used, included body-powered, myoelectric, cosmetic, and hybrid.
Heading toward the Next Level
The US Department of Defense (DoD) and the US Department of
Veterans Affairs (VA), in cooperation with various universities and
other research facilities, have brought a formidable array of
talent, funds, and collaboration to bear on improving prosthetics.
Besides osseointegration, some of the other areas being intensely
studied are CAD/CAM, prosthetic componentry, platform technology,
neuroprosthetics, and postprosthetic amputee rehabilitation. What
is discovered and developed can benefit not only active military
personnel and veterans, but also civilian populations.
The VA’s Journal of Rehabilitation Research & Development
(JRRD), May/June 2004, pointed out, “With continued military
efforts abroad and expected increases in diabetes here at home,
amputation is quickly becoming one of the more prevalent
disabilities among the veteran population.”
Among the partnering universities and medical centers are the
University of Utah and the George E. Wallen Department of Veterans
Affairs Medical Center, where several disciplines are working
together to address current concerns with osseointegration and to
build on the pioneering work done by Branemark. The focus is on the
twin issues of improving design and fighting infection.
“I know from our 25 years of implant design with our surgeon,
Dr. Aaron Hofmann, one of the top five people in the world in
implant design, that we will develop a better implant design and
better science and testing before we use these implants on these
soldiers and airmen who come back with limbs blown off,” said Roy
D. Bloebaum, PhD, in an interview with The O&P EDGE.
Bloebaum is a research professor of Orthopaedics at the University
of Utah and a senior VA hospital research scientist and co-director
of the VA Bone and Joint Research Laboratory. Hofmann is chief of
Orthopaedics at the SLC Veterans Affairs Medical Center and has his
practice at the University of Utah Orthopaedic Center.
Implant Design Challenges
One design issue involves rotation, since if a threaded implant twists inside the bone, the implant may loosen from the body. The team is incorporating a design developed by Heinz Wagner of Germany for joint replacement which involves using a grid-blasted ribbed implant–rather like inserting projecting fins–which will protect the implant from rotational failure. In addition, a porous coating made from tantalum on the most distal portion of the implant will help to keep the implant from failing axially. Bloebaum explains that tantalum has a Velcro® effect which allows the implant to achieve initial fixation. When the tantalum-coated implant is inserted into the bone, it interlocks with the bone, giving immediate axial stability, so that the implant can’t be pulled out. The ribs in the implant design prevent torsional motion as the bone heals around the implant. For instance, if the amputee were to kick a football, the leg would not fall off and the implant come flying out. Tantalum also is highly corrosion-resistant and does not react with body fluids.
In osseointegration, two interlocks occur, Bloebaum explained.
The first is mechanical, rather like driving a nail into wood. The
second interlock is initiated by the osteoblasts, which are
bone-forming cells. When the osteoblasts begin remodeling and
growing, the bone will actually start growing toward the surface of
the implant touching the bone within a six-to-nine-month period,
thus helping to adhere the implant to the skeleton.
The Utah team also is roughening the surface of the implant to a
greater degree than does Branemark. “Rougher surfaces provide
better bone contact; for reasons we don’t know yet, bone likes
roughened textures and doesn’t like smooth,” Bloebaum said. He
theorizes that a smooth surface allows blood to pool at the
interface and form a fibrous membrane which does not allow the bone
to burrow through and attach. However, a rougher surface prevents
blood pooling and formation of fibrous tissue, so the bone can
adhere better to the implant surface.
Overload Protection
Another important design strategy the Utah team is investigating is an overload protection device much like a ratchet. For instance, if an amputee were to twist a leg mountain climbing or running, the device would ratchet so that perhaps the foot would no longer be facing ahead but would be at a 90 degree angle. But the bone-implant interface would not break, and the limb would remain stable.
Such an overload protection device would be adjustable,
depending on the activity level of the amputee. The amputee’s
activity level is affected by the length of the residual limb bone.
“The more bone you have, the bigger the implant can be, and the
more loads you can take–it’s that simple,” said Bloebaum.
Infection Control
A frog is a prince in disguise when it comes to fighting
infection. In 1987, the National Institutes of Health (NIH)
discovered that the secretions produced from the African clawed
frog’s skin contains a type of antibiotic known as magainin, which
protects the frog from bacteria that is found in ponds and puddles;
one promising avenue is synthesizing the frog’s bacteria-fighting
subproteins. Magainins are part of a family of antibiotics that
help heal wounded skin rapidly.
Heading up the VA/University of Utah infection team is John
Hibbs Jr., MD, a renowned infectious disease expert, who is working
with this and other infection prevention strategies.
Implant design itself is an important component of infection
control. Bloebaum noted that Branemark now reflects muscle away
from the bone and the skin adheres to bone, which restricts the
skin motion around the abutment exiting the residual limb. “By
preventing motion at the skin abutment interface, bacteria have
limited ability to access the vulnerable tissues below the skin
implant interface,” Bloebaum said. However, bacteria do sometimes
get past this small skin barrier, possibly causing osteomyelitis
and other deep infection. The implant then can function as a growth
plate to allow a biofilm to form which perpetuates the bacteria
problem. Although antibiotics may kill the bacteria on the surface,
the bacteria underneath are still there. “It’s like just mowing off
the weeds, rather than killing the roots. So you have to keep this
biofilm from forming,” Bloebaum explained. The University of
Utah/VA team is creating some models to test Branemark’s concept,
since it does reduce chances of infection.
Other members of the VA/University of Utah team include Ken
Backus, PhD, and Steve Jacobsen, PhD, designer of the Utah Arm.
Timeline
Bloebaum estimated that his team is perhaps three to four years
away from incorporating their research into the manufacture of
devices which can be implanted into humans. These surgeries could
possibly be undertaken at the Walter Reed Army Medical Center
(WRAMC), Washington DC, or Brooke Army Medical Center (BAMC), San
Antonio, Texas. Currently the team is focusing on transfemoral
amputation strategies; as design and infection control principles
are established, they will progress to transtibial and upper-limb
amputations. “Basically our strategy is to do above-knee first,
then below-knee, then the upper limb, starting with the humerus,
since that is the easiest,” says Bloebaum. “The forearm is more
complicated because now you are dealing with two bones that can
pronate and supinate. But we have teams set up; we are progressing
methodically, determining and establishing our general principles,
then we can design the implants based on these principles. However,
we have to coordinate with the prosthetic design group. Again the
overload protection device has to be designed for the different
levels of amputation–forearm and humerus.”
Neuroprosthetics
Bloebaum is excited about the work of Richard Norman, PhD, in
neuroprosthetics, who is working on chips that can be implanted in
the brain and transmit information to the osseintegrated prosthesis
via telemetry. The prosthesis could be moved simply by thought. “It
would be fantastic if they could pull that off,” Bloebaum
enthused.
Bloebaum noted that, although they still wouldn’t duplicate the
complexity and ability of the original model humans were born with,
all these team efforts toward meeting the prosthetic challenge are
coming together to give a higher quality of life to amputees than
is currently possible.





